MYMD-1® Supera-CBD
MyMD has been featured by multiple media outlets following its merger with Akers Biosciences.
MyMD is a clinical-stage pharmaceutical company focused on the development of drug products that represent a transformation in the targeting of aging and age-related diseases; autoimmune diseases; and chronic pain, anxiety and sleep disorders. Our team has partnered with academic founders and key opinion leaders to identify synthetic therapeutics, including the first drug being designed to significantly increase human lifespan.
MyMD is commercializing two broad drug platforms, each with its own distinct indications. MYMD-1® is a novel therapeutic shown to positively impact multiple conditions related to immunometabolic dysregulation. Supera-CBD is a synthetic cannabidiol that offers better bioavailability and potency than botanicals. Both are protected by a robust, worldwide patent portfolio.
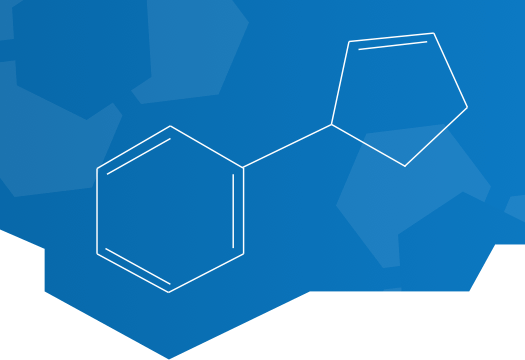
MYMD-1® is a clinical-stage, synthetic plant alkaloid that has been shown to ameliorate immunometabolic dysregulation targeting immune cell metabolism. Its mechanism of action and efficacy in autoimmune diseases, such as Multiple Sclerosis EAE (MS) and thyroiditis, and aging mouse models have been studied through MyMD’s collaboration with several academic institutions.
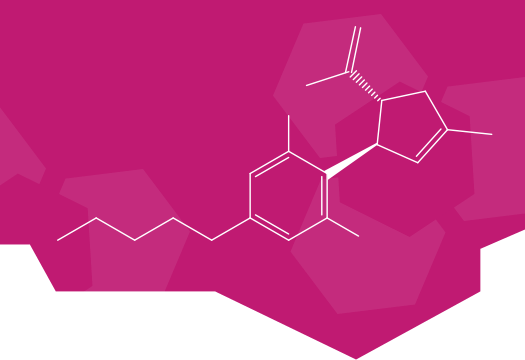
Supera-CBD is a synthetic, preclinical cannabidiol derivative that has been demonstrated to effectively target CB2 receptors. As a new molecular entity (patent pending), Supera-CBD addresses the unmet need for a synthetic CBD with enhanced bioavailability and potency so physicians may treat patients with an FDA approved drug rather than botanicals that contain impurities.
To deliver on its mission, MyMD has assembled a superb team of clinicians, experienced drug developers, US FDA / regulatory experts, patent attorneys, and business executives. The team is drawn from a variety of institutions including but not limited to Organon, Regeneron, IQVIA (formerly Quintiles), Bain & Company, and Banner Witcoff. We continue to add clinical and scientific talent to support our research and the discovery of new therapies.
About Us